I. Background Introduction:
With the rapid development of peptide drugs and other technologies, traditional amide synthesis systems still face key technical bottlenecks such as difficulty in controlling side reactions and maintaining chiral purity. Therefore, developing new strategies for efficient and highly selective amide synthesis is of great significance and application value.
II. β-Acyloxyalkenylamides (AAAs): From "High-Temperature" Intermediates to "Popular" Reagents
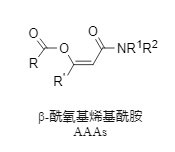
β-Acyloxyalkenylamides (Figure 1) are a class of vinyl ester compounds with high acylation reactivity. However, since their discovery, they have only been obtained through a demanding and complex five-step synthetic process. Furthermore, the key alkyne intermediate has poor stability, requiring treatment at -50 to -20°C and is difficult to store, severely limiting their large-scale preparation and application.
Figure 1
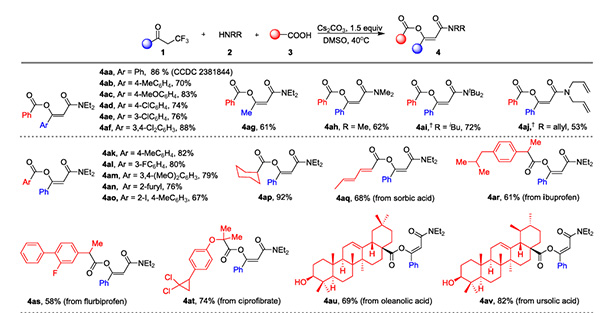
In response to this long-standing problem, Professor Wan Jieping's team recently made a significant breakthrough, developing a simple and mild method for synthesizing β-acyloxyolefin amides. Using α-trifluoromethyl ketone, secondary amines, and carboxylic acids as raw materials, and under the promotion of Cs2CO3, a continuous defluorination process was used to efficiently construct β-acyloxyolefin amides with diverse structures (Figure 2). This method has a wide range of substrate applicability, and can successfully convert ordinary carboxylic acids, protected amino acids, and bioactive molecules.
Figure 2
III. Applications: The Versatile Applicator of β-Acyloxyolefin Amides
1. Amides and Peptides
(1) Amide Synthesis
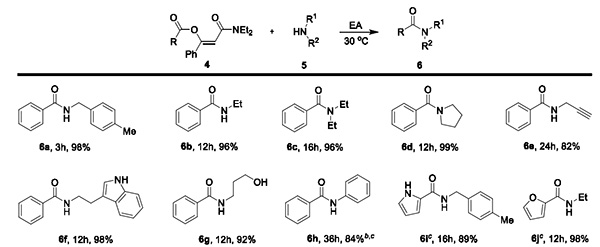
The β-acyloxyolefin amides synthesized using the new method show significant advantages in amide preparation. Under mild reaction conditions (ethyl acetate, 30℃), aliphatic amines (primary amines, secondary amines) and aromatic amines can all be obtained in high yields of the corresponding amides (Figure 3), and good compatibility is shown for β-acyloxyolefin amides with different structures.
Figure 3
(2) Peptide Synthesis
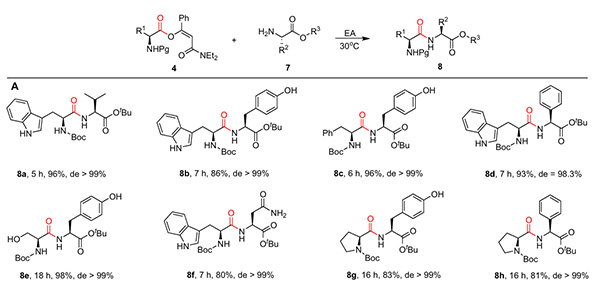
In addition, the team also explored the application of β-acyloxyolefin amides in dipeptide synthesis (Figure 4). Using amino acid-derived β-acyloxyalkenyl amides as raw materials, amidation reactions with different amino acids not only yielded the target dipeptide in excellent yields but also showed almost no racemization, demonstrating its unique advantage in chiral preservation.
Figure 4
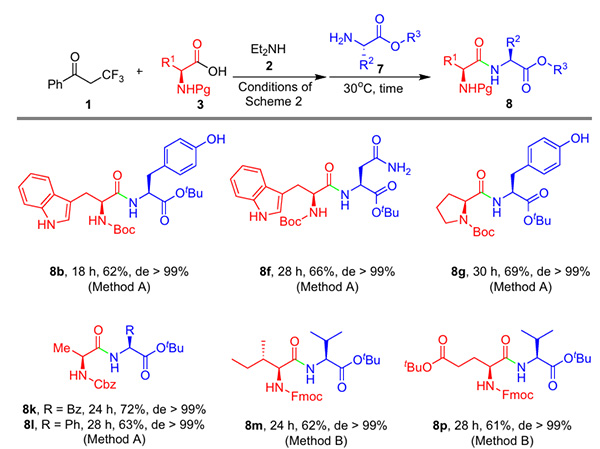
shows that, to further explore the application of β-acyloxyalkenyl amides in peptide synthesis, the team developed a one-pot two-step synthesis strategy (Figure 5): using α-trifluoromethylacetophenone, diethylamine, and N-protected amino acids as starting materials, β-acyloxyalkenyl amides were first generated through a three-component reaction. Without separation and purification, they were directly coupled with amino acid esters to obtain the corresponding dipeptide in moderate to good yields, while maintaining chiral integrity throughout the process, significantly improving synthesis efficiency.
Figure 5
(3) Racemization experiment
To objectively evaluate the racemization inhibition ability of β-acyloxyalkenyl amides in peptide synthesis, the team compared the reaction of β-acyloxyalkenyl amides with DCC and HBTU under the same conditions. The results showed that the reaction involving DCC exhibited significant racemization (dr=97.6), while the performance of β-acyloxyalkenyl amides was comparable to that of HBTU (dr>99).
2. Esters Synthesis
This strategy can also be extended to the synthesis of esters, but compared to amines, the reaction conditions are relatively harsh, requiring the reaction to be carried out at 100℃ with the participation of DBU. Under these conditions, the ester yield of the reaction with benzyl alcohol was 94%, and the ester yield of the reaction with phenol was 64% (Figure 6).
Figure 6
IV. Overall Conclusion: β-Acyloxyalkenyl Amides - A New Synthetic Tool
In summary, the new method for synthesizing β-acyloxyalkenyl amides developed by Wan Jieping's team has broad substrate applicability, successfully solving the long-standing synthetic problem in this field and realizing the transformation from "difficult to access" to "conveniently available". These compounds exhibit outstanding performance in amide and peptide synthesis, offering advantages such as mild conditions, simple operation, and good stereoselectivity, without the need for additional additives. In particular, the developed one-pot two-step synthesis strategy improves step economy and provides new synthetic approaches for peptide and drug synthesis.
Company Introduction:
Suzhou Highfine Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to global pharmaceutical R&D and manufacturing companies. Its products are mainly used in peptide, nucleotide, and pharmaceutical synthesis, covering a wide range of products including condensing agents for specialty amide bond formation, protecting agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents. Currently, it has developed and produced over 1500 products.
After more than two decades of unremitting efforts and accumulation, Highfine Biotech has continuously deepened its expertise in the global peptide synthesis reagent field and has now developed into a leading enterprise with extensive customized product coverage and significant large-scale production advantages, capable of meeting the specific needs of various customers. We sincerely invite customers interested in this product to contact us for further information and to discuss cooperation opportunities.
References:
[1] Cao, SF.; Guo, HJ; Wan, JP, et al. β-Acyloxyl alkenyl amide synthesis via multiple defluorination: α-Trifluoromethyl ketone–amine as synergystic peptide coupling reagent [J]. Sci. Adv., 2025, 11.
[2] Cao, Sufang. One-step synthesis of β-acyloxyl alkenyl amide based on CF bond activation and its application in synthesizing amides and peptides [D]. Jiangxi Normal University, 2025.