3/19/2026
Amide bonds are ubiquitous key structural units in peptide drugs, small molecule drugs, and functional materials. Previously, we introduced several studies on the amidation reactions of amines and carboxylic acids, providing rich synthetic ideas for acid-amine condensation. Today, we turn our attention to a new approach. Batra, S.'s team reported a novel amide synthesis method that enables the one-step direct reaction of nitro compounds (or nitroso compounds) with aryl α -ketocarboxylic acids to prepare amides.
Amide bonds are ubiquitous key structural units in peptide drugs, small molecule drugs, and functional materials. Previously, we introduced several studies on the amidation reactions of amines and carboxylic acids, providing rich synthetic ideas for acid-amine condensation. Today, we turn our attention to a new approach. Batra, S.'s team reported a novel amide synthesis method that enables the one-step direct reaction of nitro compounds (or nitroso compounds) with aryl α -ketocarboxylic acids to prepare amides.
The highlight of this research is that it demonstrates an innovative one-step method for the direct synthesis of amides in an aqueous medium without the need for transition metals and reducing agents. This method is simple to operate and operates under mild conditions. It has a wide range of applicable substrates, is environmentally friendly, and provides a new option for the green synthesis of amide compounds.
I. Traditional Approach
In traditional methods, the synthesis of amides from nitro compounds typically requires first reducing the nitro group to an amino group, followed by an amidation reaction with an activated carboxylic acid or in the presence of a condensing agent. This approach is not only cumbersome but also prone to generating byproducts.
Batra, S.'s team took a different approach, cleverly utilizing a free radical mechanism to achieve the direct amidation of nitro compounds (or nitroso compounds). The core of their method lies in using potassium persulfate in an alkaline aqueous solution to generate sulfate radicals, which then abstract hydrogen atoms from aryl α-ketocarboxylic acids, decarboxylating them to generate the crucial benzoyl radical , thereby constructing the amide bond .
II. Optimal Reaction Conditions and Substrate Universality
In the research of Batra, S.'s team, the reaction conditions required an aqueous system, and both the initiator and the base had to be potassium salts. The optimal reaction conditions were: acetonitrile /water (1:1) as the solvent, potassium carbonate (1.5 equivalents) as the base , potassium persulfate (3.0 equivalents) as the initiator , and nitro compounds (or nitroso compounds, 1.0 equivalents) and aryl α-ketocarboxylic acids (2.1 equivalents) reacting at 80°C to yield the corresponding amides . Furthermore, the study found that the successful reaction was highly dependent on the use of potassium salts .
This scheme demonstrates good substrate applicability and is suitable for the following types of substrates.
(1) 3-Nitroisoxazole substrates: Regardless of whether the aromatic ring has an electron-donating group (such as methyl) or an electron-withdrawing group (such as cyano), the reaction proceeds smoothly and yields the corresponding amide product in excellent yield. Various substituted aryl α- ketocarboxylic acids can also participate in the reaction well.
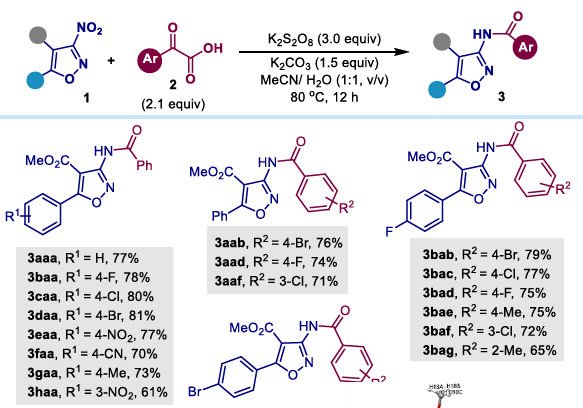
However, isoxazole substrates require an electron-withdrawing group at the 4-position for conversion to occur; amidation cannot be achieved in the absence of substitution.
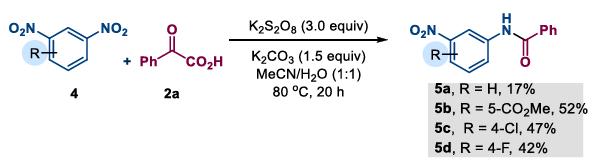
(2) 1,3-Dinitrobenzene substrates: The team also explored some amidation reactions of 1,3-dinitrobenzene and its derivatives, obtaining the corresponding N-(3-nitrophenyl)benzamide products in yields of 17-52%. This method provides a new route for the direct derivatization of polynitro compounds.
(3) Nitrosyl substrates: Based on a deep understanding of the mechanism, researchers have successfully extended this strategy to nitrosyl compounds. Under the same conditions, nitrosyl compounds and aryl α- ketocarboxylic acids directly yield a series of structurally diverse amides in moderate to high yields. This transformation is applicable to a variety of substituted nitrosyl aromatics, heterocyclic nitrosyl compounds, and aliphatic nitrosyl compounds.
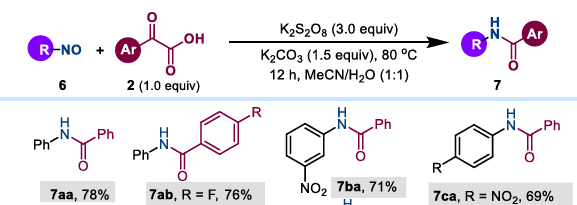
Even more interestingly, the study found that N-benzoylphenylhydroxylamine can be directly converted into amides in aqueous solution in the presence of potassium persulfate without the need for a base. In other words, as long as the structure of the reaction substrate meets a certain stage of the mechanism, amides can be synthesized using this method.
III. Reaction Mechanism: Radical-Driven Amide Reaction
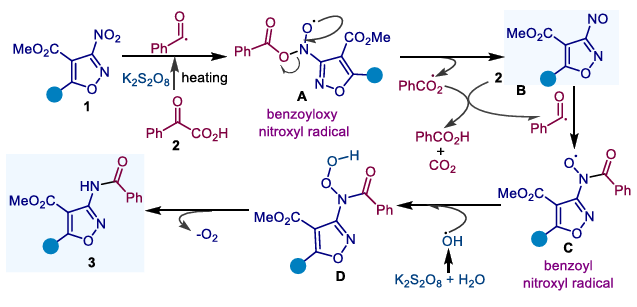
The team proposed the following reaction mechanism: Taking 3-nitroisoxazole substrates as an example, firstly, under a high-temperature alkaline environment, potassium persulfate decomposes to generate sulfate radicals, which then abstract hydrogen atoms from aryl α-ketocarboxylic acids and undergo decarboxylation to generate benzoyl radicals. Subsequently, this radical attacks the nitro group in the nitroaromatic hydrocarbon, abstracting an oxygen atom to generate benzoyloxy radicals and nitrosoaromatic hydrocarbon intermediates. The benzoyloxy radical reacts with another molecule of aryl α-ketocarboxylic acid to regenerate benzoyl radicals. Next, a second benzoyl radical combines with a nitrosoaromatic hydrocarbon to form a benzoylnitroso radical intermediate. Finally, this radical combines with hydroxyl radicals generated within the system, undergoing a deoxygenation process to ultimately generate the target amide product.
IV. Synthesis, Application, and Summary
In summary, this study developed a direct amidation method that is mild, simple to operate, requires no transition metals or reducing agents, and is environmentally friendly. Utilizing a free radical transformation process, it successfully achieved the efficient and green conversion of 3-nitroisoxazoles, 1,3-dinitrobenzenes, and nitroso compounds into amides, providing an attractive new route for the synthesis of related amide compounds and offering important theoretical basis and practical examples for the field of green synthetic chemistry .
Company Introduction:
Suzhou Haofan Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to pharmaceutical R&D and manufacturing companies worldwide. Its products are mainly used in the synthesis of peptides, nucleotides, and pharmaceuticals, covering a wide range of products including condensing agents for specialty amide bonds, protective agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents. To date, it has cumulatively developed and produced over 1,500 different products.
After more than two decades of unremitting efforts and accumulation, Haofan Biotech has continuously cultivated its expertise in the global peptide synthesis reagent field. It has now developed into a leading enterprise with extensive customized product coverage and significant advantages in large-scale production, capable of meeting the specific needs of various customers. We sincerely invite customers interested in this product to contact us to learn more about product details and explore cooperation opportunities .
References:
[1] Decarboxylative/OxidativeAmidationofArylα‑KetocarboxylicAcidswith Nitroarenes and Nitroso Compounds in Aqueous Medium.
DOI: 10.1021/acs.orglett.0c03666
Please fill out the form below and our sales team will be happy to assist you with a quote on peptide synthesis reagents.