1. Background Introduction
From traditional reagents to a constant stream of novel designs, they have greatly driven the innovation of peptide synthesis technology. New-generation reagents have significantly improved synthesis efficiency and purity, enabling the construction of peptide bonds to be more efficient and precise, while also providing essential tools for life sciences and new drug research and development.
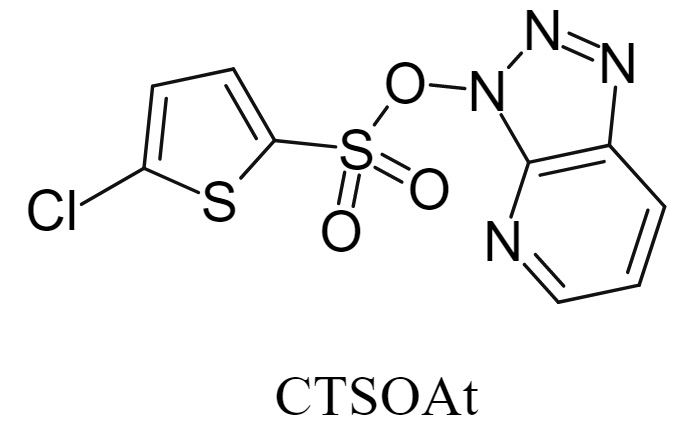
2. CTSOAt:
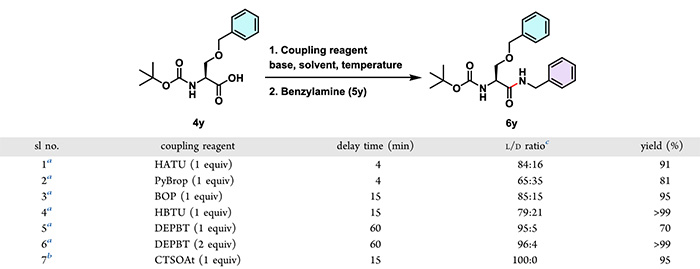
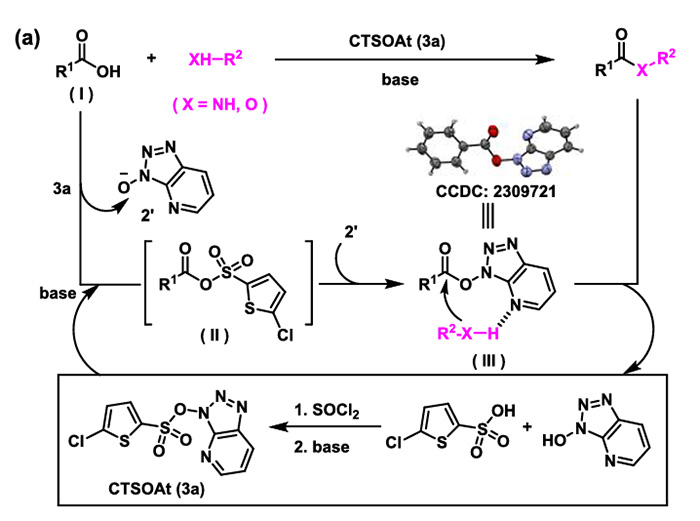
3H-[1,2,3]triazolo[4,5-b]pyridin-3-yl 5-chlorothiophene-2-sulfonate (abbreviated as CTSOAt, whose structure is shown in the figure below) is a novel multifunctional coupling reagent that can be used to promote the formation of C-O, C-N, and C-C bonds and is recyclable. In terms of stability, this reagent can be stably stored at 0°C for half a year. In addition, experimental evidence shows that the key linkage in the CTSOAt molecule is via an S-O bond rather than an S-N bond, and the former has a lower bond energy, resulting in a more stable overall structure. Furthermore, compared to HOAt, which has potential thermal stability hazards, CTSOAt has been proven to possess excellent thermal stability through DSC and TGA tests.
![CTSOAt CTSOAt]()
Notably, long-term validation has demonstrated that coupling reagents based on the HOAt structure generally exhibit excellent racemization-inhibiting capabilities. CTSOAt not only retains the structural moiety of HOAt, but its thiophene structure can also form favorable interactions with carboxylic acids, further facilitating the retention of configuration.
3. Applications
As a novel, multifunctional and high-efficiency coupling reagent, CTSOAt can not only promote amidation and esterification reactions, but also participate in Suzuki reactions to realize the conversion of carboxylic acids to ketone compounds.
1. Amides and Polypeptides
(1) Amide Synthesis
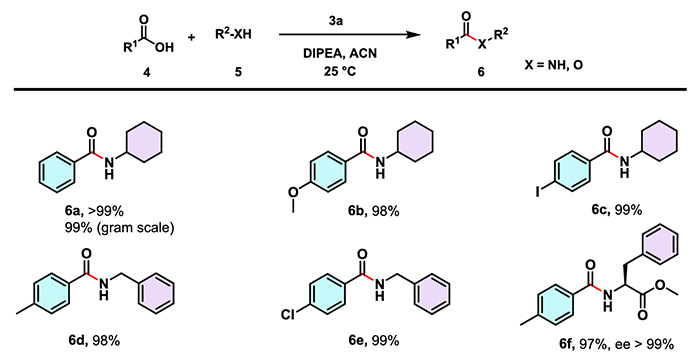
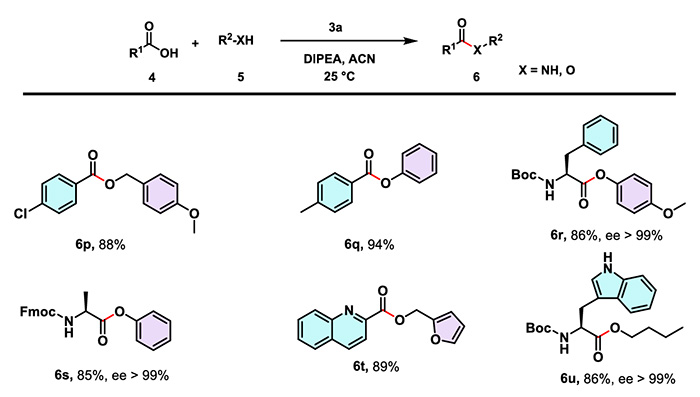
The research team led by Mandal Bhubaneswar systematically evaluated the applicability of CTSOAt in amide synthesis, covering aromatic acids (with electron-withdrawing and electron-donating groups), (long-chain) fatty acids, as well as amine compounds such as aromatic amines, aliphatic amines and sterically hindered amines. Experimental results showed that the yields of target compounds generally exceeded 90%. Even for substrates containing base-sensitive Fmoc-protected groups, CTSOAt could still efficiently catalyze the reaction to afford the corresponding amides smoothly.
(2) Peptide Synthesis
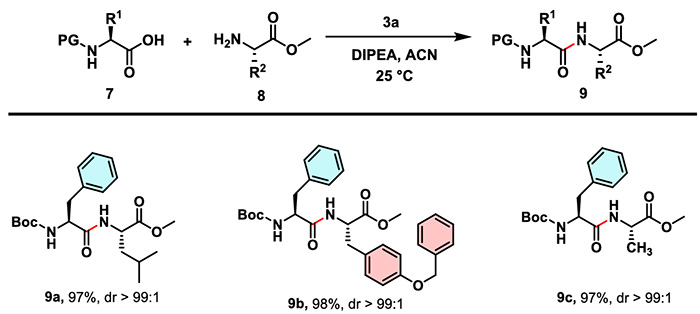
Under the same conditions, CTSOAt has been successfully applied to the synthesis of dipeptides and tripeptides containing various protecting groups and sterically hindered moieties. The reactions deliver high yields and excellent performance in stereoconfiguration retention, which fully demonstrates its applicability in the assembly of complex peptide segments.
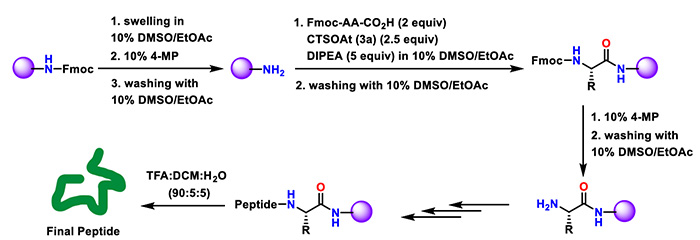
(3) Solid-Phase Synthesis
In solid-phase synthesis, the green solvent system of 10% DMSO/ethyl acetate was adopted, and peptide sequences were synthesized on Rink amide MBHA resin following the Fmoc/t-Bu strategy. CTSOAt achieved satisfactory yields across all tested sequences. Specifically, in the synthesis of KLVFF—a core fragment analog of amyloid-β (Aβ)—the yield reached 56%, whereas the yield of HATU under the same conditions was only 27%.
(4) Drug ModificationCTSOAt can also be applied to the amidation structural modification of various drug molecules, such as gemfibrozil and indomethacin. These cases further verify its practical value in reactions involving sterically hindered and low-reactivity amines.
(5) Racemization TestTo thoroughly evaluate the racemization-inhibiting capability of CTSOAt, the research team led by Mandal Bhubaneswar conducted a horizontal comparison with currently mainstream coupling reagents (e.g., DEPBT, HBTU). The results demonstrated that CTSOAt enables the production of target compounds at a yield of 95% within a short period, with no racemic products detected.
2. Ester Synthesis
CTSOAt is equally applicable to the synthesis of ester compounds. Compared with amine compounds, the yields of esters synthesized from alcohols with weaker nucleophilicity are generally lower than those of amides. Even so, the yields can still exceed 80%, and the product exhibits excellent stereoconfiguration retention.
The reaction mechanism of such coupling reactions mediated by CTSOAt is as follows:
Under the action of a base, the carboxylic acid attacks CTSOAt to form a mixed anhydride intermediate. Subsequently, the oxygen anion in the HOAt moiety attacks the mixed anhydride, yielding an activated OAt ester. Finally, the alcohol/amine initiates a nucleophilic attack to afford the corresponding ester and amide. During the reaction, 5-chlorothiophene-2-sulfonic acid and HOAt are generated as by-products. After treatment with thionyl chloride, these two by-products enable the recovery of CTSOAt with a 50% recovery rate.
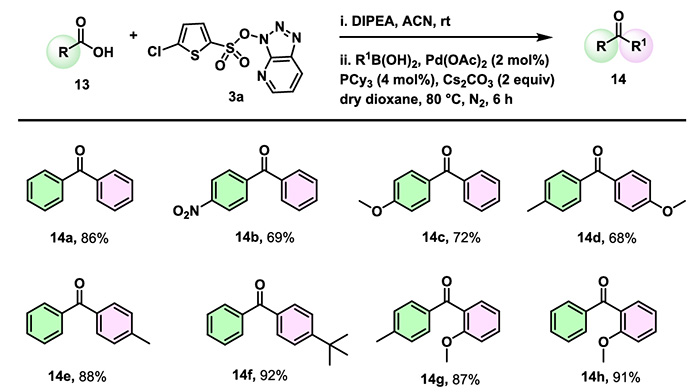
3. Ketone Synthesis
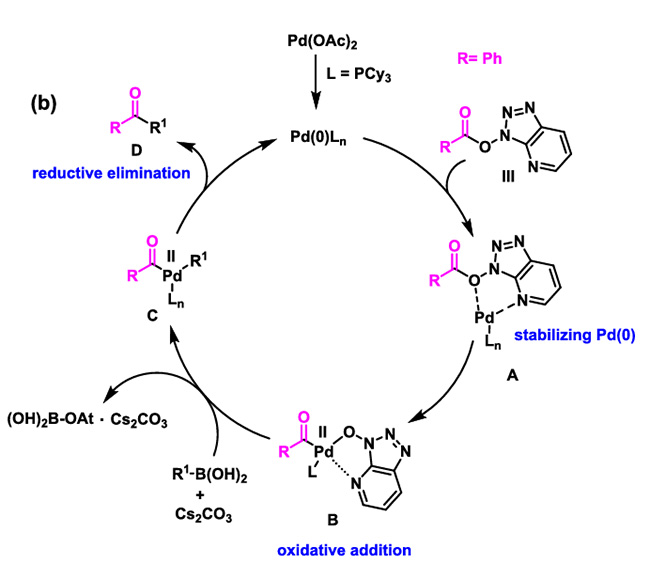
Beyond conventional coupling reactions, CTSOAt can also participate in Suzuki coupling reactions to enable the conversion of carboxylic acids into ketone compounds. Carboxylic acids and boronic acids bearing electron-withdrawing groups, electron-donating groups or sterically hindered substituents all afford the target compounds with moderate to good yields.
The specific reaction mechanism is as follows: Under Pd catalysis, the conversion of carboxylic acids to ketones is achieved through processes including oxidative addition, transmetalation, and reductive elimination.
4. Overall Conclusions
CTSOAt is a novel and recyclable coupling reagent with excellent functional group tolerance and steric hindrance tolerance. This reagent can not only efficiently promote the synthesis of amides and esters and significantly inhibit the occurrence of racemization, but also demonstrate broad application prospects in drug molecule modification and solid-phase synthesis. Furthermore, it can participate in Suzuki coupling reactions to enable the conversion of carboxylic acids into ketone compounds. In summary, the development of CTSOAt provides a new and powerful tool for synthetic chemistry and related fields.
About Highfine Biotech
Suzhou Highfine Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise dedicated to supplying specialty raw materials for pharmaceutical R&D and manufacturing enterprises worldwide.
Our products are mainly applied in fields such as peptide synthesis, nucleotide synthesis and pharmaceutical synthesis, covering a wide range of categories including specialty coupling reagents for amide bond formation, protecting reagents, linkers, protein cross-linkers for antibody-drug conjugates (ADCs), molecular building blocks, liposomes and phosphorus reagents. Up to now, we have R&D and production experience of more than 1,500 kinds of products in total.
After more than two decades of persistent efforts and accumulation, Highfine Biotech has been deeply engaged in the global peptide synthesis reagent sector. It has now evolved into a leading enterprise with extensive custom product coverage and distinct large-scale production advantages, capable of meeting the specific needs of various customers.
We sincerely invite customers interested in our products to contact us for further details and discussion on potential cooperation opportunities.
References:
[1] Roy, S.; Rahaman, S. R.; Sarkar, A., Mandal, B. Sulfonate Derivative of HOAt (CTSOAt) as a Coupling Reagent for the Construction of C−N, C−O, and C−C Bonds[J]. J. Org. Chem. 2025, 90, 34, 12061–12079.