I. Background Introduction:
With the increasing importance of amide bonds in pharmaceutical molecules, fine chemicals, and agrochemicals, especially given the unique properties of fluorine-containing structures in various fields, higher demands are placed on condensation reagents in terms of synthesis efficiency and applicability. Therefore, developing multifunctional condensation reagents has significant research and application value.
II. Diethylaminosulfur trifluoride (DAST):
DAST (Figure 1) is a commonly used deoxyfluorinating agent that can convert alcohols, aldehydes, and ketones into their corresponding monofluorinated and geminal difluorinated compounds. Furthermore, DAST can also convert carboxylic acids into acyl fluorides, demonstrating potential condensation applications. Based on this, Natte's team developed a method for synthesizing challenging amides using DAST in dichloromethane with equimolar ratios of carboxylic acids and amines as raw materials and DAST as the condensing agent. The condensation reaction can be completed at room temperature without the need for additional alkali.
Figure 1
III. Applications:
1. Synthesis of Fluorinated Carboxylic Acid Amides from Difluorobromoacetic Acid
Difluorobromoacetic acid is used as the starting carboxylic acid. It undergoes amidation reactions with different amine compounds (primary and secondary amines) to obtain the corresponding amide products in moderate to good yields. Some products can be obtained directly by crystallization, simplifying post-processing. This method is applicable to aliphatic and aromatic amines (including electron-rich and electron-deficient amines), demonstrating good substrate applicability. However, the reaction yield is lower for sterically hindered substrates.
Figure 2
2. Synthesis of Long-Chain Fatty Acid Amides from p-Trifluoromethylaniline
Long-chain fatty acid amides play an important role in multiple fields due to their unique properties, and market demand continues to grow. Traditional methods require converting long-chain fatty acids into acyl chlorides or using traditional condensation reagents such as DCC for synthesis, resulting in cumbersome post-processing. However, the synthesis of amides using DAST as a condensation reagent is simple in reaction purification, and most products can be obtained directly by crystallization. The operation is convenient, and the yield is moderate.
Figure 3
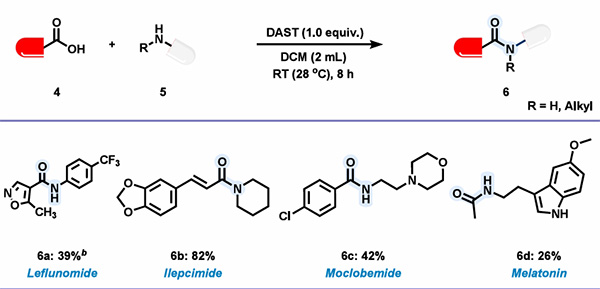
3. Drug Molecule Synthesis:
The DAST-mediated amidation reaction exhibits good functional group tolerance. Natte's team further investigated the effect of this reagent in drug synthesis. As shown in the figure below, drugs such as leflunomide, eleximide, moclobemide, and melatonin were synthesized using corresponding carboxylic acids and amines, with separation yields of 26-82% for the target products.
Figure 4
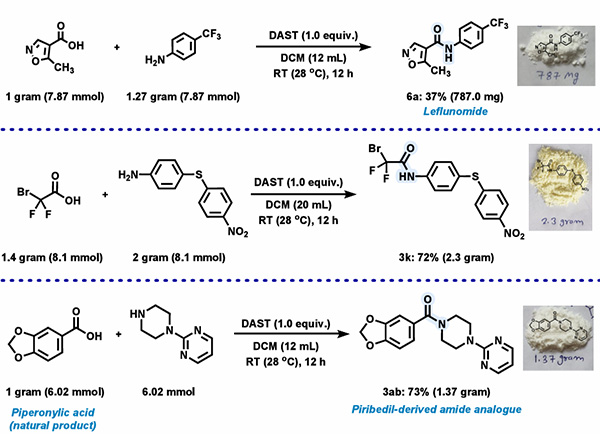
4. Gram-Scale Experiment:
To verify the scalability of this method, Natte's team conducted gram-scale experiments. The results showed that the scale was comparable to that of the 0.5 mmol scale experiment, indicating that the method has good scalability.
Figure 5
IV. Reaction Mechanism
The DAST-mediated amidation reaction mechanism is as follows:
Pathway A: DAST reacts with carboxylic acids to form intermediate A and releases HF. Subsequently, intermediate A loses fluoride ions to form a more reactive intermediate B, which then undergoes nucleophilic addition with fluoride ions to obtain an acyl fluoride intermediate and releases intermediate D.
Pathway B: Intermediate D also reacts with carboxylic acids to obtain intermediate E, and simultaneously reacts with HF to release sulfur dioxide and diethylamine to obtain the acyl fluoride intermediate.
Two pathways work synergistically to generate an acyl fluoride intermediate, which then reacts with an amine compound under the promotion of diethylamine to obtain the corresponding amide product. However, it was found in experiments that the released diethylamine may also participate in the amidation reaction, leading to side reactions.
Figure 6
V. Overall Conclusion:
In summary, the Natte team has developed a simple, non-alkaline, and convenient post-processing method for amide synthesis. This method uses DAST to promote amide bond formation, has broad substrate applicability and synthetic applicability, and provides new ideas for the synthesis of amide bonds in multiple fields.
Company Introduction:
Suzhou Highfine Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to global pharmaceutical R&D and manufacturing companies. Its products are mainly used in the synthesis of peptides, nucleotides, and pharmaceuticals, covering a wide range of products including condensing agents, protecting agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents for specialty amide bond formation. Currently, it has cumulatively developed and produced more than 1500 products.
After more than 20 years of unremitting efforts and accumulation, Highfine Biotech has continued to cultivate the global peptide synthesis reagent field and has now developed into a leading enterprise with extensive customized product coverage and significant advantages in large-scale production, which can meet the specific needs of various customers. We sincerely invite customers who are interested in this product to contact us to learn more about the product details and explore cooperation opportunities.
References:
[1] Kumawat, S.; Wohlrab, S.; Natte, K., et al. DAST Enabled Synthesis of Fluorinated Amides and Fatty Acid Amides Including Drugs under Ambient Conditions [J]. Org. Lett., 2025, 27, 32, 8829–8834.