I. Background Introduction:
However, this efficient cyclical synthetic strategy of protection-condensation-deprotection significantly increases the number of peptide synthesis steps, reagent consumption, and cumbersome operations. Therefore, developing a method that directly achieves amino acid cross-condensation reactions without protection is the best solution.
Recently, Yamamoto, H.'s team proposed a highly efficient and green synthetic route based on silicon reagents: modifying amino acids with silicon reagents to make them electrophilic and nucleophilic, and directly condensing them without the use of traditional protecting groups to efficiently construct peptide bonds. This method not only simplifies the synthetic steps but also demonstrates excellent practicality.
II. Principle Introduction:
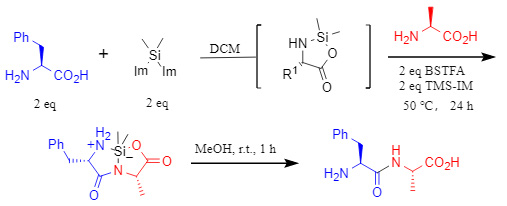
The core of this study lies in the synergistic effect of two types of silane reagents to achieve highly selective amide bond formation between unprotected amino acids. Specifically, one amino acid molecule reacts with bis(1-imidazolyl)dimethylsilane in dichloromethane to yield an electrophilic silicon-containing five-membered cyclic intermediate, which can act as an acyl donor to undergo subsequent reactions with other nucleophiles to generate the corresponding products (see Figure 1); another amino acid molecule forms an amino acid-TMS ester as a nucleophile under the combined action of N-trimethylsilylimidazolium (TMS-IM) and N,O-bis (trimethylsilyl) trifluoroacetamide (BSTFA).
Figure 1
In this system, BSTFA primarily acts as a silylation reagent, while TMS-IM plays a dual role: firstly, it reacts with amino acids to enhance their solubility in dichloromethane; secondly, it assists the silylation process. The synergistic effect of these two agents significantly improves reaction selectivity, ultimately yielding a silane-cyclic dipeptide with the correct sequence through the electrophilic and nucleophilic reactions.
Furthermore, the resulting silane-cyclic dipeptide exhibits good stability under normal conditions, requiring only rapid treatment with methanol at room temperature to obtain the unprotected dipeptide, facilitating subsequent extension or purification (Figure 2).
Figure 2
III. Applications:
1. Wide substrate applicability and excellent stereoselectivity
This synthetic strategy exhibits good compatibility with amino acids of different structures (Figure 3). Whether it is an amino acid with a sterically hindered side chain or a phenylalanine derivative with different electronic properties, the target product can be obtained in high yield (most exceeding 80%), and the stereoconfiguration of the amino acid can be well maintained (dr > 20:1). Even for phenylglycine (Phg), which is prone to racemization, its stereoconfiguration can still be maintained, demonstrating the superiority of this method in terms of stereoselectivity.
Figure 3
2. Efficient elongation from dipeptide to polypeptide
The silane-cyclic dipeptide intermediates obtained through this synthetic strategy are stable, easy to manipulate, and readily convertible into unprotected dipeptides, while also providing a unique platform for peptide chain elongation. Both the N-terminus and C-terminus can be directly coupled without the involvement of traditional condensation reagents to achieve peptide chain elongation, effectively avoiding the formation of oxazolone and the resulting epimerization problems commonly encountered in long peptide chain synthesis.
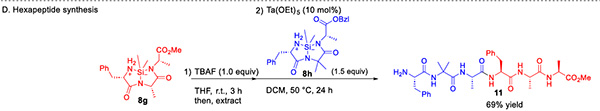
This study further demonstrates the application potential of this strategy in the construction of complex sequences. Through the direct condensation between silane-cyclic tripeptides, hexapeptide and heptapeptide sequences were successfully constructed (Figure 4), with the heptapeptide backbone widely found in numerous drug molecules such as smegglutide and telposide. This "convergent" synthetic strategy, which assembles target sequences by directly linking peptide segments, significantly simplifies the synthetic process and provides a novel approach for the preparation of complex peptides and peptide drugs.
Figure 4
IV. Overall Conclusion:
In summary, this study developed a method for the formation of unprotected chemically selective peptide bonds between amino acids based on silicon reagents, and achieved aggregated peptide synthesis. This method not only significantly reduces the number of synthetic steps and reagent consumption, but also provides a convenient route for the preparation of target peptides under mild conditions. This breakthrough provides a novel synthetic route for the field of peptide synthesis and is expected to promote the further development of peptide drugs and related research.
Company Introduction:
Suzhou Highfine Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to global pharmaceutical R&D and manufacturing companies. Its products are mainly used in peptide, nucleotide, and pharmaceutical synthesis, covering a wide range of products including condensing agents for specialty amide bond formation, protecting agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents. Currently, it has developed and produced over 1500 products.
Through more than two decades of unremitting efforts and accumulation, Highfine Biotech has continuously deepened its expertise in the global peptide synthesis reagent field and has now developed into a leading enterprise with extensive customized product coverage and significant advantages in large-scale production, capable of meeting the specific needs of various customers. We sincerely invite customers interested in our products to contact us for further information and to discuss cooperation opportunities.
References:
[1] Peptide Bond Formation Between Unprotected Amino Acids: Convergent Synthesis of Oligopeptides. DOI: https://doi.org/10.1021/jacs.4c08049