I. Background Introduction
Currently, various methods for synthesizing esters/amides from carboxylic acids have been developed, involving various chemical reagents, catalysts, and reaction media. Among them, titanium tetrachloride (TiCl4), in addition to its conventional applications such as the removal of N-protecting groups, has previously been reported for the synthesis of amides and esters, but this usually requires relatively high temperatures. In recent years, the conditions for TiCl4-mediated condensation reactions have been further optimized, enabling the synthesis of amides and esters at essentially room temperature.
II. TiCl4-Mediated Amide Synthesis
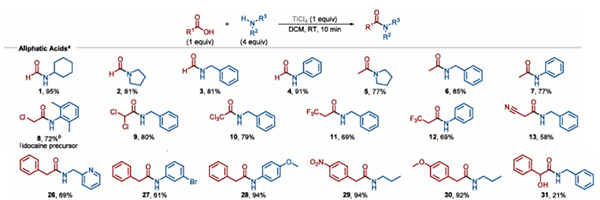
The Ramachandran, PV team systematically studied the application of TiCl4 in amide synthesis: at room temperature, carboxylic acids were dissolved in dichloromethane, 1.0 eq TiCl4 was added, and 4.0 eq amine was slowly added dropwise. The reaction was carried out for 10 min to obtain the corresponding amide compound.
This method is applicable to the reaction of aliphatic carboxylic acids with aliphatic amines or aromatic amines (Figure 1), and can obtain the target amide in moderate to good yields. The nucleophilicity of the amine compound has a certain influence on the reaction yield.
Figure 1. TiCl4-mediated condensation reaction of aliphatic carboxylic acids with amines
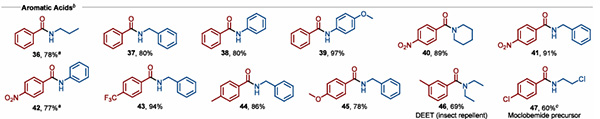
For aromatic carboxylic acids, the yield of amides synthesized under the above standard conditions is low. Studies have found that adding an additional 4.0 eq of pyridine to the system can significantly promote amide formation, with yields reaching up to 97% (Figure 2).
Figure 2. TiCl4-mediated condensation reaction of aromatic carboxylic acids with amines
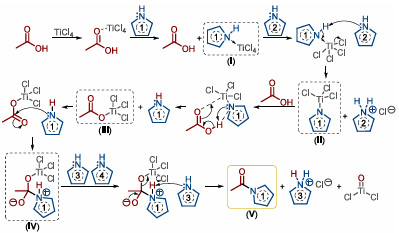
To determine the role of TiCl4 in the reaction system, the team used NMR to detect the reaction progress and proposed the following reaction mechanism (Figure 3): TiCl4 first coordinates with the carbonyl oxygen of the carboxylic acid, then forms a TiCl4-amine complex (I) with the added amine and releases the carboxylic acid; under the action of another amine molecule, it deprotonates to form a trichloroamine titanium intermediate (II), which is the rate-determining step of the reaction; then the carboxylic acid reacts with trichloroamine titanium to generate an activated carboxylic acid ester (III); the amine attacks the activated ester in a nucleophilic addition reaction, and with the assistance of the additional amine, deprotonates, finally yielding the target amide product (V).
Figure 3. Mechanism of TiCl4-mediated amide condensation reaction
In addition, the team expanded upon the active intermediate of trichloroamine-titanium, successfully achieving its conversion with substrates such as acetophenone and DMF to obtain the corresponding amine and amide compounds.
III. TiCl4-mediated ester synthesis
The Leggio, A. team explored the application of TiCl4 in ester synthesis: at room temperature, carboxylic acids were dissolved in dichloromethane, 3.0 eq TiCl4 was added, and after reacting for 20 minutes, 3.0 eq primary alcohol was added. The reaction was continued at room temperature for 8-16 h to obtain the corresponding ester compounds.
This method is suitable for (long-chain) fatty acids and aromatic carboxylic acids, and the reaction with primary alcohols yields the target ester in over 70% yield (Figure 4). However, the reaction is less effective for sterically hindered carboxylic acids, resulting in a decrease in reaction yield.
Figure 4. TiCl4-mediated condensation reaction of carboxylic acids with primary alcohols
For alcohols that readily form stable carbocations, the above conditions will produce the corresponding chlorinated derivatives. Studies have found that replacing the reaction solvent with the nonpolar solvent n-hexane can effectively suppress the formation of carbocations, directing the reaction towards ester formation. Phenolic compounds, due to their low reactivity, require reflux under toluene conditions to successfully yield the corresponding esters (Figure 5).
The team proposed the following ester bond formation mechanism: carboxylic acids react with TiCl4 to generate activated ester intermediates of the carboxylic acid; then, alcohols/phenols attack the active ester in a nucleophilic addition reaction to obtain the target ester product. The entire process can proceed smoothly without the participation of a base.
Figure 5: TiCl4-mediated condensation reaction of carboxylic acids with phenols and secondary alcohols.
IV. Outlook
In summary, TiCl4-mediated condensation reactions provide a mild and practical reaction route for the synthesis of esters and amides. The reactions can proceed smoothly at room temperature, are simple to operate, and exhibit good adaptability to various carboxylic acids and nucleophiles. With further research, TiCl4 is expected to play an increasingly important role in the field of organic synthesis.
V. Company Introduction
Suzhou Highfine Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to global pharmaceutical R&D and manufacturing companies. Our products are mainly used in peptide, nucleotide, and pharmaceutical synthesis, covering a wide range of products including condensing agents for specialty amide bond formation, protective agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents. Currently, we have developed and produced over 1500 different products.
Through more than two decades of unremitting efforts and accumulation, Highfine Biotech has continuously deepened its expertise in the global peptide synthesis reagent field and has now developed into a leading enterprise with extensive customized product coverage and significant advantages in large-scale production, capable of meeting the specific needs of various customers. We sincerely invite customers interested in our products to contact us for further information and to discuss cooperation opportunities.
References:
[1] Rapid, Room-Temperature Amidation via Tandem Titanium Amido Complex and Titanium Carboxylate Intermediates. DOI: 10.1021/acs.orglett.5c03336
[2] Titanium Tetrachloride-Assisted Direct Esterification of Carboxylic Acids.
DOI: 10.3390/molecules29040777