3/13/2026
Amide bonds are not only the basic structural unit of peptide molecules, but also the core structural unit of many natural products and drug molecules. With the increasing complexity of bioactive molecule design, the cumbersome protection-deprotection steps in traditional peptide synthesis strategies have become a bottleneck restricting synthesis efficiency, increasing the length of the synthetic route and potentially causing side reactions and decreased yields.
I. Background Introduction:
Amide bonds are not only the basic structural unit of peptide molecules, but also the core structural unit of many natural products and drug molecules. With the increasing complexity of bioactive molecule design, the cumbersome protection-deprotection steps in traditional peptide synthesis strategies have become a bottleneck restricting synthesis efficiency, increasing the length of the synthetic route and potentially causing side reactions and decreased yields.
In recent years, the direct condensation strategy of unprotected amino acids has gradually become a research hotspot. This method aims to simplify the synthesis process, reduce costs, and improve atom economy. In this field, following their previous development of silane-mediated direct condensation between amino acids, Yamamoto, H.'s team has recently turned their attention to organometallic reagents — trimethylaluminum (Me3Al ) —to explore its application in peptide synthesis.
II. Trimethylaluminum:
Trimethylaluminum is a highly reactive Lewis acid with strong electrophilicity and methylation capabilities. As an organoaluminum reagent, it can participate in various types of reactions, exhibiting unique advantages, particularly in activating carboxyl groups and promoting amidation reactions. However, the fact that trimethylaluminum reacts with water naturally makes it somewhat hazardous and requires a higher level of handling expertise.
Prior to Yamamoto, H.'s team's research on trimethylaluminum-mediated peptide synthesis, there had been several studies on the use of trimethylaluminum for amide synthesis.
III. Early Exploration:
1. Pfizer's research team previously reported the use of trimethylaluminum to promote the direct condensation of unprotected carboxylic acids and amine compounds. The mechanism of action is to achieve the construction of amide bonds by forming aluminate intermediates .
In the substrate suitability study, 4-phenylbenzoic acid was used as the fixed carboxylic acid and amidation reactions were carried out with different types of amine compounds. Under the action of trimethylaluminum, primary amines, secondary amines and aromatic amines were all obtained with excellent yields to the target amides.
Furthermore, using 4-phenylbenzylamine as the fixed amine component, reactions with different types of carboxylic acids were conducted, and it was found that aromatic acids generally exhibited better activity than fatty acids.
2. Martin, SF's team developed a method for promoting the reaction of amino acid esters with free amino acids to form peptide bonds using trimethylaluminum. This method uses carboxylic acid esters or N-protected amino acid esters as electrophiles to directly couple with free amino acids or dipeptides, achieving good yields. However, when attempting to further extend the peptide chain to synthesize tripeptides and tetrapeptides, significant racemization occurs, limiting its application in the synthesis of long peptides.
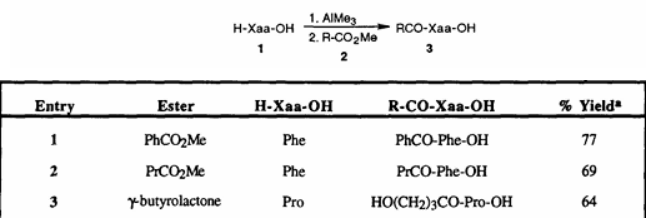
While the above studies have verified the potential and application value of trimethylaluminum in amidation, they generally have limitations in the synthesis of long peptide chains and cannot meet the needs of preparing complex peptide structures.
IV. One-pot peptide synthesis strategy :
Yamamoto, H.'s team proposed a one-pot peptide synthesis strategy based on a five-membered ring mechanism. This method fully utilizes the multifunctionality of trimethylaluminum, which both activates the carboxyl group and coordinates the nucleophilic attack of the amino group, achieving efficient and highly stereoselective peptide synthesis.
1. One-pot reaction mechanism
Its mechanism of action is as follows: First, trimethylaluminum reacts with free amino acids to activate the carboxyl group and release methane; then it reacts with an intramolecular amino group to form a five-membered ring intermediate A and releases a second molecule of methane; then, under the catalysis of another molecule of trimethylaluminum, the newly added amino acid ester attacks intermediate A to undergo a ring-opening reaction, giving intermediate B; finally, acid treatment releases the amino group, yielding the dipeptide product.
If it is necessary to further extend the peptide chain, the above cycle can be repeated based on the synthesis of dipeptides, or Fmoc-protected aminoacyl chloride can be introduced for end capping in the last step, thereby precisely controlling the peptide chain length.
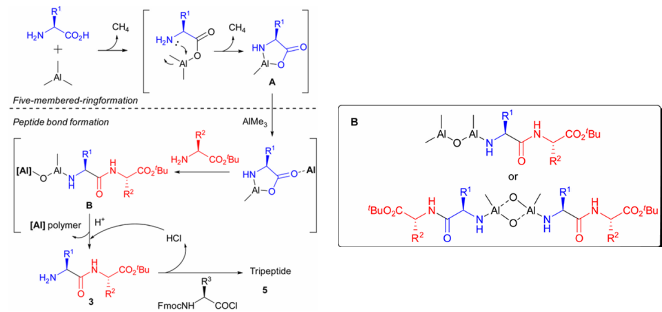
2. Optimization of reaction conditions
The team used tripeptide synthesis as a model reaction to explore reaction conditions. They eventually determined that dichloromethane was used as a solvent, and that free amino acids and trimethylaluminum were premixed at low temperature (0 °C ) to activate the carboxyl group. Then, the tripeptide was subjected to amidation reaction with amino acid methyl ester at room temperature and acid treatment with pentyl acid to ensure that the amino group was completely free. Finally, the tripeptide was obtained by reacting with Fmoc to protect the amino acyl chloride.
3. Excellent functional group compatibility and stereoselectivity
This synthetic strategy exhibits excellent functional group compatibility, is tolerant to amino acids containing side chains, as well as amino acids containing ester groups, ether bonds, and sulfur-containing groups, and has a high reaction yield. No obvious racemization was observed during the entire synthesis process, and it has good stereoselectivity.
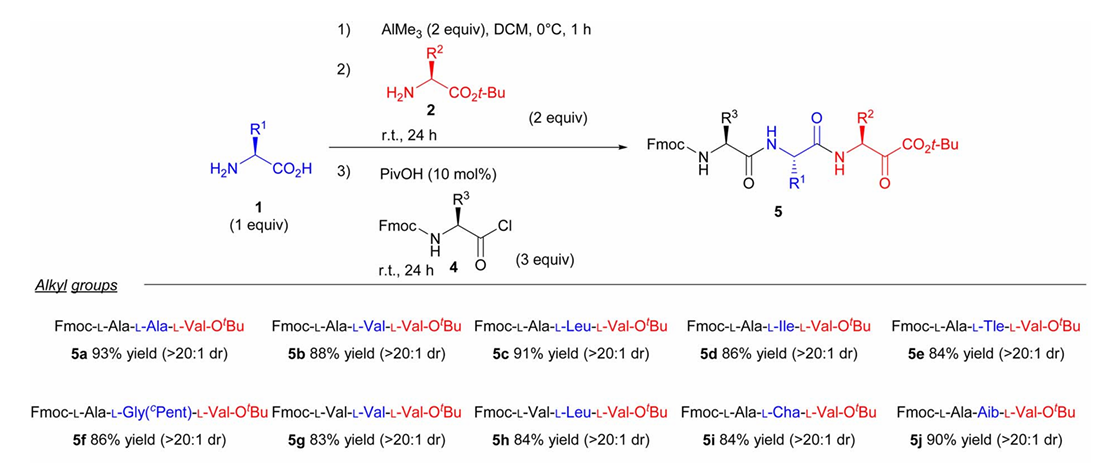
Building upon this foundation, the team further explored the stepwise synthesis of tetrapeptides and pentapeptides, maintaining a synthesis yield of around 60%, demonstrating good scalability. It is worth noting that the study found the synthesized peptide chain length and yield to be significantly affected by the product solubility.
V. Overall Conclusion:
In summary, trimethylaluminum is an inexpensive, readily available, efficient, and low-racemic condensation reagent. Its use eliminates the need for pre-activation of the carboxylic acid, significantly reducing synthetic steps and reagent consumption. It also exhibits good functional group compatibility and provides a convenient route for the preparation of target peptides under mild conditions. This breakthrough offers a novel approach to the synthesis of peptide drugs and functional peptide materials.
Company Introduction:
Suzhou Haofan Biotech Co., Ltd. (Stock Code: 301393.SZ), founded in 2003 and headquartered in Suzhou High-tech Zone, is a national high-tech enterprise providing specialty raw materials to pharmaceutical R&D and manufacturing companies worldwide. Its products are mainly used in the synthesis of peptides, nucleotides, and pharmaceuticals, covering a wide range of products including condensing agents for specialty amide bonds, protective agents, linking agents, protein cross-linking agents for antibody-drug conjugates, molecular building blocks, liposomes, and phosphorus reagents. To date, it has cumulatively developed and produced over 1,500 different products.
After more than two decades of unremitting efforts and accumulation, Haofan Biotech has continuously cultivated its expertise in the global peptide synthesis reagent field. It has now developed into a leading enterprise with extensive customized product coverage and significant advantages in large-scale production, capable of meeting the specific needs of various customers. We sincerely invite customers interested in this product to contact us to learn more about product details and explore cooperation opportunities .
References:
[1] Trimethylaluminium-Facilitated Direct Amidation of Carboxylic Acids.
DOI: 10.1055/s-0030-1260982
[2] Application of AlMe3-mediated amidation reactions to solution phase peptide synthesis.
DOI: 10.1016/s0040-4039(98)00071-9
[3] Trimethylaluminum-mediated one-pot peptide elongation.
DOI: 10.1039/d3sc00208j
Please fill out the form below and our sales team will be happy to assist you with a quote on peptide synthesis reagents.